In these Combustion and Flame notes for Class 8 Science, students will gain a deeper understanding of one of nature’s most captivating processes. They will appreciate the scientific principles that govern combustion, the characteristics of flames, and the significance of fire safety in daily life. So, let’s embark on this enlightening journey into the world of combustion and flame, unraveling the mysteries of fire along the way.

Fire has been a vital discovery in the history of humankind, providing warmth, light, and a means for cooking food. But have you ever wondered what exactly happens when something burns? How does fire produce heat and light? The answers lie in the fascinating phenomenon of combustion and flame. Let’s get started with combustion and flame notes.
Combustion and Flame Notes – Class 8 Science
Throughout these notes, we will explore the different aspects of combustion. We will understand the conditions required for combustion and flame to take place, including the presence of fuel, oxygen, and a source of ignition. We will also study the various types of fuels, both natural and man-made, and their role in the process of burning.
Combustion and flame are integral to our daily lives, from the warmth of a cozy fire to the energy that drives our modern world. By delving into the intricacies of this phenomenon, we gain a deeper understanding of its principles and the importance of responsible handling.
What is Combustion?
A chemical process in which a substance reacts with the oxygen of the air to give heat and light is called combustion. And the substance which undergoes combustion is called a combustible substance or a fuel. The fuel may be a solid, liquid, or gas.
During combustion, light is also given off sometimes, either as a combustion and flame or as a glow. Example: Magnesium burns in the air and combines with oxygen to form magnesium oxide, liberating heat, and light.

Combustible and Non-combustible Substances
- Combustible substances (Fuel): Substances that undergo combustion or burn are called combustible substances. Examples: Paper, cloth, cooking gas (LPG), CNG, kerosene oil, wood, charcoal, etc. You can buy some of these online.
- Non-combustible substances: Substances that do not undergo combustion are called non-combustible substances. Examples: Stones, cement, bricks, soil, sand, water, iron nails, etc.

Conditions Necessary for Combustion and Flame
- Presence of a combustible substance (a substance which can burn).
- Presence of a supporter of combustion (like air or oxygen).
- Heating the combustible substance to its ignition temperature.
Ignition Temperature
Ignition temperature is the lowest temperature at which a substance catches fire. That means if the temperature of a substance is less than the ignition temperature required, then the substance will not catch fire.
For example, Petrol, Diesel can catch fire with a very low flame because their ignition temperature is low. In comparison, wood will only catch fire when we put it in contact with fire for some time. It needs some time to achieve the minimum temperature required to catch the fire.
Inflammable Substances
The substances which have very low ignition temperatures and can easily catch fire with a flame are called inflammable substances. Examples: Petrol, alcohol, and Liquefied Petroleum Gas (LPG).
Matchsticks
- Matchstick or a match is a type of tool for starting a fire.
- Earlier, they were made up of a mixture of antimony trisulphide, potassium chlorate, and white phosphorus with some glue.
- Starch was applied on the head of the match made up of some suitable wood.
- Due to the heat of friction, white phosphorus got ignited. But it was dangerous for the workers in matchstick manufacturing and users.
- Later, the mixture contained only antimony trisulphide and potassium chlorate. The rubbing surface of the match was covered with powdered glass and red phosphorus (less dangerous).
- When the match strikes against the rubbing surface, some of the red phosphorus is converted into white phosphorus and reacts with potassium chlorate to produce enough heat to ignite the antimony trisulphide.
Types of Combustion
The combustion is categorized into the following three types. These are Rapid Combustion, Spontaneous Combustion, and Explosion.
- Rapid Combustion: If the substance burns rapidly and produces heat and light. Such combustion is called Rapid Combustion. For example, burning the gas stove in the home with the use of a lighter or matchstick.
- Spontaneous Combustion: When the substance burns into flames suddenly on its own without any known reason, this is called Spontaneous Combustion. For example, sudden fires in the coal mines. This might be due to heat or sunlight.
- Explosion: When a sudden reaction turns into heat, light, and sound. Such combustion is very fast and produces large amounts of heat, light, and sound. This is called an explosion. For example, igniting a firecracker will instantly become heat, light, and sound. The explosion can even happen because of the pressure on the cracker.
How do We Control Fire?
Things required to start the fire are Fuel (combustible substance), Air (to supply Oxygen), and Heat (raising the temperature to reach Ignition temperature). Hence, to control the fire, we need to control the above conditions.
1. Remove the Fuel
Fuel is a food for fire. So, when a fire starts, all the combustible materials should be removed so the fire does not spread.

2. Cut off the Air Supply
- Carbon dioxide is the best extinguisher for fires involving electrical equipment and inflammable materials such as petrol.
- Being heavier than oxygen, carbon dioxide covers the fire like a blanket. If a person catches fire, an actual physical blanket can also be used as it cuts off the air supply.
- As a result, the contact between the fuel and oxygen is cut off, and the fire is controlled.
3. Remove the heat or reduce the temperature
- The most common fire extinguisher is water, but it works for ordinary fires.
- When water is thrown on a burning substance, it gets cooled below its ignition temperature and stops burning.
- If electrical equipment is on fire, water may conduct electricity and can give an electric shock to people involved in firefighting.
- Water is also not suitable for fires involving oil and petrol. Because water is heavier than oil, it sinks below the oil, and the oil keeps on burning at the top.
Carbon Dioxide
Carbon Dioxide is one of the best fire extinguishers. Let’s review its characteristics in controlling a fire.
- Carbon dioxide is stored at high pressure as a liquid in cylinders.
- On releasing it from the cylinder, it expands enormously in volume and cools down.
- It forms a blanket around the fire and brings down the temperature of the fuel. So, it is an excellent fire extinguisher.
- Carbon Dioxide is also produced by releasing a lot of dry powder of chemicals such as sodium bicarbonate or potassium bicarbonate. Near the fire, these chemicals give off Carbon Dioxide.
Learn more about Combustion and Flames with Important Questions on Vector Tutorials App.
Flame
In the combustion process, light is also released along with heat. This visible, gaseous part of the fire is called flame. Kerosene oil, wax, LPG etc., are some of the substances which give flame on burning.
Structure of Flame
If you look closely at a flame, you can see its colours and different layers of flame. These layers form the structure of flame, which has three parts, i.e., Outer Zone, Middle Zone, and Inner Zone.
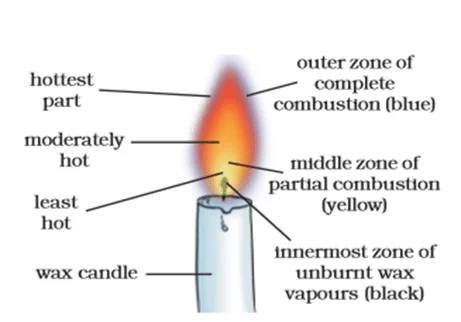
- Outer Zone: This is the outermost part of a flame and is blue in colour. This part is the hottest in a flame where complete combustion takes place.
- The outermost zone is also called the “oxidation zone” or “blue zone.”
- The high temperature in this zone leads to the emission of blue-colored light, indicating the presence of a higher energy level.
- Middle Zone: This is the middle zone of a flame and is moderately hot. This part is yellow in colour and shows partial combustion. It is also known as the “luminous zone” or “yellow zone.”
- This zone is responsible for the visible light emitted by the flame.
- It contains combustible gases, such as carbon monoxide and hydrogen, undergoing partial combustion.
- The incomplete combustion results in the release of energy in the form of light, giving the flame its characteristic yellowish glow.
- The temperature in this zone is higher than in the innermost zone but lower than in the outermost zone.
- Inner Zone: This is the innermost zone of the unburnt substance and the least hot part of a flame. This is the central region of the flame, often referred to as the “dark zone” or “non-luminous zone.”
- It contains unburned or partially burned fuel particles.
- The temperature in this zone is relatively low, as the fuel particles are not fully combusted due to a lack of sufficient oxygen.
- The primary role of this zone is to supply combustible gases to the outer zones for further combustion.
Please note that the structure of a flame may vary depending on the fuel being burned and the availability of oxygen. The size and shape of the flame can also be influenced by factors such as airflow, fuel composition, and the presence of impurities.
Fuels
The substance which goes under combustion is called fuel. Fuels can be solid, liquid, or gas. Petrol, wood, charcoal, etc., are some examples of fuel. Understanding fuels is necessary to study Combustion and Fuels notes.
- Good fuel doesn’t cost much and is always cheap.
- A good fuel is easily available.
- It should generate a large amount of heat.
- Such type of fuel should burn at a slow rate, and combustion must be controllable.
- There should not be any release of gases that can harm the environment.
- There should be the complete combustion of that fuel (i.e., no unburnt substance left).

Ideal Fuel
The fuel which satisfies all of the above conditions is termed ideal fuel. It should be cheap, easily available, have high calorific value, and complete combustion and flame with proper ignition temperature. There is probably no such fuel which is ideal.
Fuel Efficiency
Fuel efficiency refers to the measure of how effectively a fuel is converted into useful energy, usually in the form of work or heat, with minimal waste or loss. It is an essential aspect of energy consumption and has implications for environmental impact, cost savings, and sustainability.
The amount of heat energy produced on the complete combustion of 1kg of fuel is called its calorific value. This is expressed in a kilojoule per kg unit (kJ/kg). Different fuels produce different amounts of heat on burning.
| Fuel | Calorific Value (kJ/kg) |
|---|---|
| Cow dung cake | 6000 – 8000 |
| Wood | 17000 – 22000 |
| Coal | 25000 – 30000 |
| Petrol | 45000 |
| Kerosene | 45000 |
| Diesel | 45000 |
| Methane | 50000 |
| CNG | 50000 |
| LPG | 55000 |
| Biogas | 35000 – 40000 |
| Hydrogen | 150000 |
Importance of Fuel Efficiency
- Environmental Impact: Improving fuel efficiency helps reduce greenhouse gas emissions and minimize the environmental impact associated with fuel consumption, such as air pollution and climate change.
- Cost Savings: Higher fuel efficiency means less fuel is required to perform a specific task, resulting in reduced fuel costs for individuals, businesses, and industries.
- Energy Conservation: Enhancing fuel efficiency contributes to the conservation of finite energy resources, promoting long-term sustainability and energy security.
Harmful Effects of Burning Fuels
- Due to the partial combustion of carbon fuels such as wood, coal, and petroleum. The unburn carbon particles become air pollutants and cause respiratory diseases such as asthma.
- This partial combustion of fuels releases Carbon Monoxide, which is a poisonous gas. If coal is burnt in a closed room, then the person sleeping in that room can be killed by this CO gas.
- Global Warming: The rise in the temperature of the atmosphere of the earth is called Global Warming. Combustion of most fuels releases CO2, which increases the concentration of CO2 in the air, and this is the biggest reason for Global Warming.

- Acid Rain: Burning of coal and diesel releases the chemicals like Sulphur Dioxide, and petrol engine release Nitrogen Dioxide. These are suffocating and corrosive gases that are very dangerous to the environment. When these oxides of Sulphur and Nitrogen dissolve in rainwater, they form acids. Such rain is called acid rain, which is much harmful to crops, buildings, and soil.
To avoid such harmful effects, CNG (Compressed Natural Gas) is being used in automobiles instead of diesel and petrol. CNG gas produces harmful products in relatively very small amounts and can be used as a cleaner fuel. You may also see some other alternatives, such as Electric vehicles and Hydrogen based vehicles, in upcoming years.
Some important points
- Fuel differs in its efficiency and cost.
- Fire can be controlled by removing one or more requirements essential for producing fire.
- Cutting off trees to obtain wood leads to Deforestation, which is quite harmful to the environment and humans.
- You should never play fire with dry grass as it catches fire immediately and can spread within minutes. Burning dry grass might be the reason for forest fire which is too difficult to control.
- With technological advancement and judicial uses of fuel, we can save fuels, increase their efficiency and keep the environment safe.
Frequently Asked Questions on Combustion and Flame (FAQs)
That was all about the Combustion and Flame notes for class 8 science. In these combustion and flame notes, we have provided the important points to cover the basics of combustion, flame, and fuels. Students are suggested to go through these combustions and flame notes a few times to clear all their doubts. Combustion is a chemical reaction that occurs when a substance combines with oxygen, releasing energy in the form of heat and light. It is an essential process for various human activities, from cooking and heating to powering engines and generating electricity.
Remember, these combustion and flame notes are just the spark to ignite your curiosity. Exploring further, conducting experiments, and observing real-life examples will deepen your understanding of the captivating world of Combustion and Flame. Stay curious, stay safe, and let the flames of knowledge illuminate your path! 🔥
It is necessary to keep in mind that fire can be both a valuable tool and a destructive force. There are some precautions and measures you should take to prevent and control fires, ensuring the safety of ourselves and our surroundings.
More notes for Class 8 Science